What Is Helium?
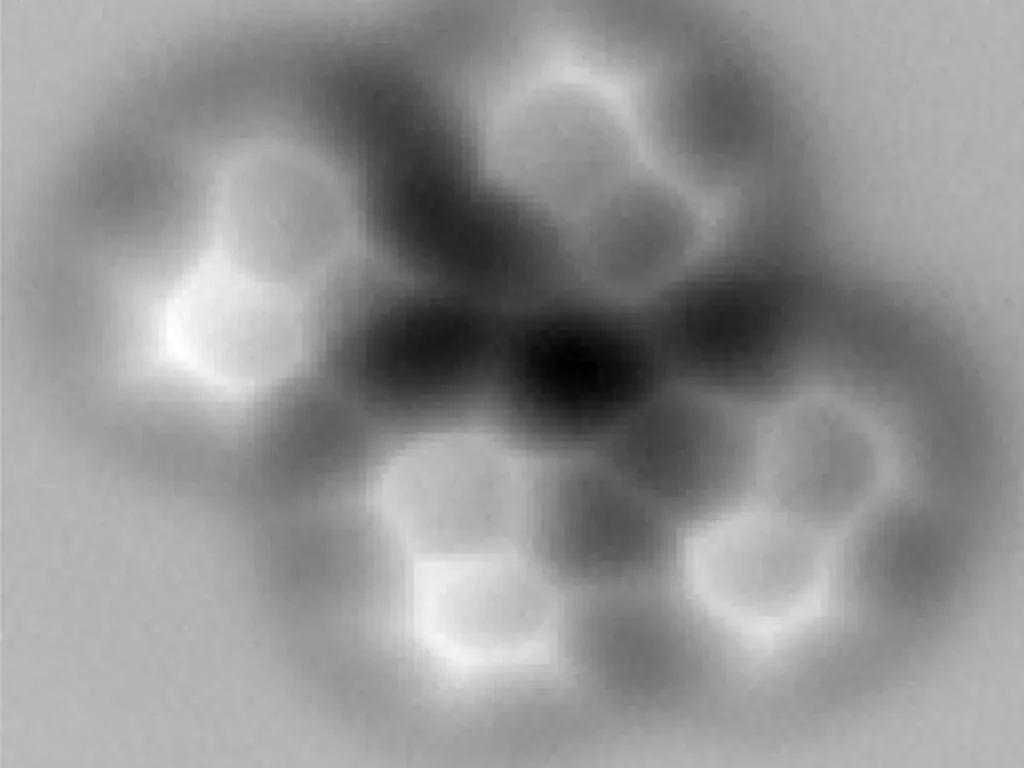
Helium is the second element on the periodic table (atomic number 2). It is a colorless, odorless, tasteless, non-toxic, and inert gas. Helium is the second most abundant element in the universe, but it is relatively rare on Earth.
A helium atom has two protons, two neutrons, and two electrons. Unlike almost every other element, helium does not combine with other atoms. It is called a noble gas because it almost never forms chemical compounds.

Interesting fact: Helium was first discovered in the Sun before it was found on Earth. Its name comes from “Helios,” the Greek word for Sun.
Key Properties
| Property | What It Means |
|---|---|
| Color | Colorless |
| Smell | Odorless |
| Taste | Tasteless |
| State at room temperature | Gas (He) |
| Reactivity | Almost zero (inert) |
| Density | Very light — second lightest after hydrogen |
| Flammability | Not flammable |
Where Do We Find Helium in Daily Life?
You encounter helium more often than you realize, mainly because of its unique physical properties.
In Balloons and Airships
Helium is lighter than air, so it makes balloons float. Unlike hydrogen, helium is not flammable, making it safe for party balloons, weather balloons, and blimps.
In Medicine
Liquid helium is extremely cold (about -269 degrees Celsius). It is used to cool the superconducting magnets inside MRI (Magnetic Resonance Imaging) machines. Without liquid helium, most MRI machines would not work.
In Scientific Research
Helium is used to cool particle accelerators and powerful microscopes to near absolute zero. It is also used as a carrier gas in gas chromatography, a technique for separating chemical mixtures.
In Industry
Helium is used as a shielding gas in welding because it does not react with the hot metal. It is also used to pressurize and purge rocket fuel tanks.
In Diving
Deep-sea divers sometimes breathe a mixture of helium and oxygen instead of regular air. Helium is less soluble in blood than nitrogen, which reduces the risk of decompression sickness (the bends). It also makes divers’ voices sound squeaky and high-pitched.
In Leak Detection
Because helium atoms are very small and inert, manufacturers inject helium into pipes or containers and use special sensors to detect tiny leaks.
Interesting Facts About Helium
- Helium is the only element that was discovered outside Earth before being found on Earth. Scientists saw it in the Sun’s spectrum in 1868, and it was found on Earth in 1882.
- Helium has the lowest boiling point of any element: minus 268.93 degrees Celsius. It remains a liquid even at absolute zero unless placed under pressure.
- Helium does not freeze solid at normal pressure. To make solid helium, you must apply about 25 times normal atmospheric pressure even at temperatures near absolute zero.
- When you inhale helium from a balloon, your voice becomes squeaky and high-pitched. This happens because sound travels faster in the lighter helium gas than in air. Warning: Inhaling too much helium can cause suffocation because it displaces oxygen.
- Helium is a non-renewable resource on Earth. It forms naturally from the radioactive decay of uranium and thorium deep underground, where it gets trapped in natural gas deposits. Once released into the atmosphere, it becomes so light that it escapes into space forever.
- The United States used to store a massive national helium reserve in Texas. This reserve once supplied much of the world’s helium.
- Helium is used to cool the Large Hadron Collider at CERN, the world’s largest particle accelerator, to temperatures colder than outer space.
- Some stars burn helium into carbon and oxygen through nuclear fusion. This process happens in older, heavier stars.
- Helium is the second most abundant element in the universe after hydrogen, making up about 24 percent of all normal matter.
- There is a type of mineral called “hélium” or uranium-bearing minerals that contain small amounts of helium trapped from radioactive decay.
A Safety Note
While helium is not toxic or flammable, breathing it directly from a pressurized tank can cause lung damage or even death. Inhaling a small amount from a balloon for a funny voice is generally safe in moderation, but inhaling too much displaces oxygen and can cause dizziness, loss of consciousness, or brain damage. Never inhale helium directly from a tank.
Summary in One Sentence
Helium is the light, inert, non-flammable gas that makes balloons float, cools MRI machines, and is the second most abundant element in the universe but a limited resource on Earth.
For Science Lovers (Quick Reference)
- Symbol: He
- Atomic number: 2
- Atomic mass: approximately 4.0026 u
- Electron configuration: 1s²
- Melting point: does not solidify at normal pressure
- Boiling point: -268.93 degrees Celsius
- Density at room temperature: 0.1786 grams per liter (air is about 1.2 grams per liter)
- Main isotopes: Helium-4 (most common), Helium-3 (rare, used in nuclear research)